Whole animal protein turnover studies
Claydon, A.J., Thom, M.D., Hurst, J.L. & Beynon, R.J. (2011) Protein turnover: measurement of proteome dynamics by whole animal metabolic labelling with stable isotope labelled amino acids. Proteomics (in press)
The measurement of protein turnover in tissues of intact animals is obtained by whole animal dynamic labelling studies, requiring dietary administration of precursor label. It is difficult to obtain full labelling of precursor amino acids in the diet and if partial labelling is used, calculation of the rate of turnover of each protein requires knowledge of the precursor relative isotope abundance (RIA). We describe an approach to dynamic labelling of proteins in the mouse with a commercial diet supplemented with a pure, deuterated essential amino acid. The pattern of isotopomer labelling can be used to recover the precursor RIA, and sampling of urinary secreted proteins can monitor the development of liver precursor RIA non-invasively. Time-series analysis of the labelling trajectories for individual proteins allows accurate determination of the first order rate constant for degradation. The acquisition of this parameter over multiple proteins permits turnover profiling of cellular proteins and comparisons of different tissues. The median rate of degradation of muscle protein is considerably lower than liver or kidney, with heart occupying an intermediate position.
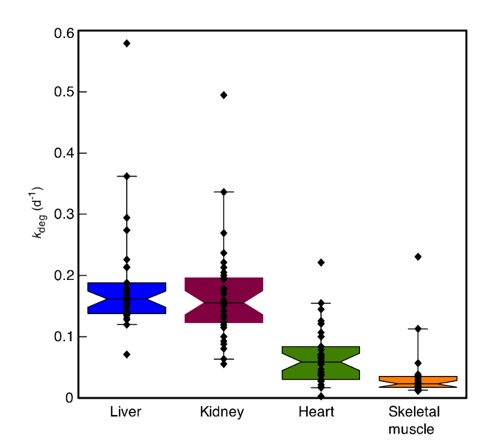
The measurement of protein turnover in tissues of intact animals is obtained by whole animal dynamic labelling studies, requiring dietary administration of precursor label. It is difficult to obtain full labelling of precursor amino acids in the diet and if partial labelling is used, calculation of the rate of turnover of each protein requires knowledge of the precursor relative isotope abundance (RIA). We describe an approach to dynamic labelling of proteins in the mouse with a commercial diet supplemented with a pure, deuterated essential amino acid. The pattern of isotopomer labelling can be used to recover the precursor RIA, and sampling of urinary secreted proteins can monitor the development of liver precursor RIA non-invasively. Time-series analysis of the labelling trajectories for individual proteins allows accurate determination of the first order rate constant for degradation. The acquisition of this parameter over multiple proteins permits turnover profiling of cellular proteins and comparisons of different tissues. The median rate of degradation of muscle protein is considerably lower than liver or kidney, with heart occupying an intermediate position.
